 Such crystalline solid compounds of anions and cations are often referred to as salts. In an ionic bond, the metal atoms release their valence electrons, which are taken up the nonmetal atoms in order to reach the noble gas configuration for each atom! Figure: Ionic bound of common salt (table slat) 
The ionic bond has special significance for ceramics. The cohesion between the metal and non-metal atoms is due to the electrostatic attraction of the resulting ions. The non-metal atom becomes a negatively charged ion ( anion) after the electrons are taken up. 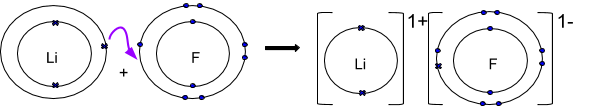
The metal atom becomes a positively charged ion ( cation) after the release of the electrons. In both cases, the noble gas configuration for the respective atoms is achieved. 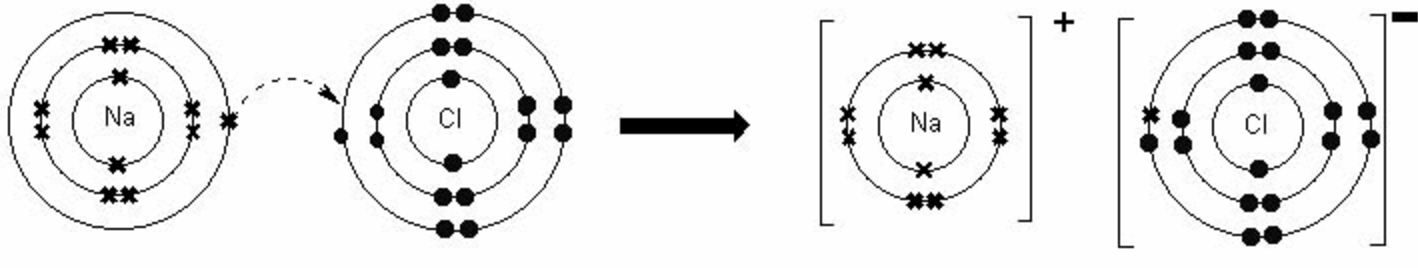
The metal atoms involved in the binding release their valence electrons, which are taken up by the nonmetal atoms. The ionic bond is the predominant type of bonding between a metal and a nonmetal. In ionic bonding, the metal atoms give off their outer electrons, which are taken up by the non-metal atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
